Relative
Dating
Prior
to the availability of radiocarbon dates (and when there is no material
suitable for a radiocarbon date) scientists used a system of relative dating.
Relative dating establishes the sequence of physical or cultural events
in time. Knowing which events came before or after others allows scientists
to analyze the relationships between the events.
For example, archaeologists might date materials based upon relative depth of burial in a site. The archaeologists record and analyze the changes in types and styles of human-made items from different levels according to the principle explained below.
General
principle
Archaeologists,
geologists, and other scientists make use of this principle when dating
sediments or layers relative to one another:
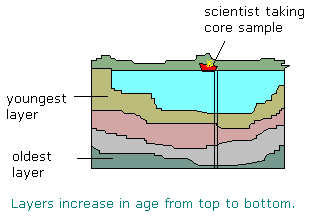
The principle of superposition states that in an undisturbed sequence of sediments, layers increase in age from top to bottom.Drawbacks of relative dating methods
Relative methods do not always reflect the true sequence of events in time. There are potential problems with relative dating.
 |
Sediment core from Moon Lake. |
Sediments are usually laid down in horizontal beds. Any observable tilting or swirling is due to disruption of the process. This should be reflected in the dating. Material that intrudes or cuts into a horizontal bed is assumed to be younger than the material that is disrupted.
Consider a lake that dries out or somehow contains older sediments that are washed into it. These sediments are deposited on younger sediments currently being deposited in the lake. The resulting sequence from the bottom-most sediments would be oldest-to- young-to- older.Relative dating methods also do not result in an absolute age - only an indication of whether items are younger or older than each other.
Radiocarbon
Dating
C14
Formation
Radiocarbon
dating is a widely used method of obtaining absolute dates on organic material. Carbon C14 is a type of carbon that undergoes radioactive decay at a known rate. Read more about its formation here.
Organic
materials that can be dated
Many
different organic (carbon containing) materials may be dated using the
radiocarbon method. Wood, seeds, hair, bone, insect remains, peat, and
charcoal are just a few of the materials that are radiocarbon dated.
How
is C14 used for dating?
All plants
and animals incorporate carbon into their tissues during their lives for
growth and energy. When an organism dies, it stops incorporating carbon
(all forms of carbon, including C14) into its structure.
The amount of radioactive carbon (C14) that had been in the organism when it was alive begins to decrease at death as it loses nuclear particles through radioactive decay. In effect, the "clock" starts ticking when death occurs.
Carbon 14 decays at a constant rate. After 5,568 years, half of the original amount of C14 in the living organism remains. In another 5,730 half of that amount will remain, and so on. In order to date a sample of unknown age, the proportion of C14 relative to C12 are carefully determined, then compared to the proportion of a modern, measured sample.
Radiocarbon dating is difficult when testing a sample that is older than about 50,000 years because the amount or remaining C14 is so small that it is very difficult to measure.
See the Web Resources list for more information on C14 dating.
|
|
Copyright © 2000 Illinois State Museum